Intraosseous Infusion - Femoral: Difference between revisions
From Protocopedia
m Treloars moved page Intraosseous Infusion to Intraosseous Infusion - Femoral: Adding Humeral Head Page |
mNo edit summary |
||
| Line 39: | Line 39: | ||
* Utilize pressure (syringe bolus, pressure bag or infusion pump) for continuous infusions where applicable. Begin infusion. Dress site, secure tubing and apply wristband as directed. Monitor EZ-IO® site and patient condition – Remove catheter within 24 hours. | * Utilize pressure (syringe bolus, pressure bag or infusion pump) for continuous infusions where applicable. Begin infusion. Dress site, secure tubing and apply wristband as directed. Monitor EZ-IO® site and patient condition – Remove catheter within 24 hours. | ||
[[Category:Procedure Guidelines]] | [[Category:Procedure Guidelines|0914.01]] | ||
Latest revision as of 17:36, 1 February 2018
Procedure Guidelines
9.14.1 INTRAOSSEOUS INFUSION - FEMORAL
The SCEMS Medical Director has approved the EZ IO device for Intraosseous (IO) access:
INDICATIONS:
- Immediate vascular access in emergencies.
- Intravenous fluids or medications are urgently needed and a peripheral IV cannot be established in 2 attempts or 90 seconds AND the patient exhibits one or more of the following:
- An altered mental status (GCS of 8 or less)
- Respiratory compromise (SaO2 90% after appropriate oxygen therapy, respiratory rate < 10 or > 40 min)
- Hemodynamic instability (Systolic BP of < 90).
- Cardiac arrest (medical or traumatic)
- Profound hypovolemia with alteration of mental status
- Burns blocking peripheral IV sites
CONTRAINDICATIONS: (If a contraindication is present evaluate the opposite leg for placement.)
- Fracture to the tibia or femur.
- Previous orthopedic procedures. (Example – knee replacement)
- An extremity that is compromised by a pre-existing medical condition. (Example – tumor or peripheral vascular disease)
- Any infection or burn* over the insertion site.
- Excessive tissue at insertion site with the absence of anatomical landmarks (consider alternate site)
- Relative contraindication. If patient has extensive burns, the site can be used if unable to obtain an IV.
PROCEDURE: EZ IO
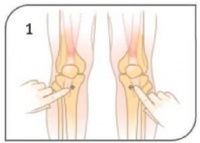
- Locate appropriate insertion site (Multiple sites are FDA cleared including - Proximal / Distal Tibia and Proximal Humerus)
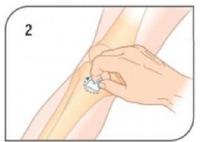
- Prepare insertion site using aseptic technique. Prepare the EZ-IO® driver and appropriate needle set
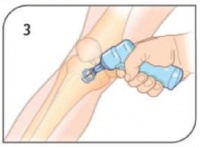
- Stabilize site and insert appropriate needle set. Remove EZ-IO® driver from needle set while stabilizing catheter hub
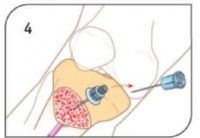
- Remove stylet from catheter, place stylet in shuttle or approved sharps container. Confirm placement.
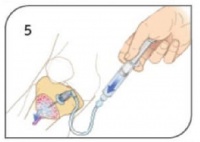
- Connect primed EZ-Connect®. Slowly administer appropriate dose of Lidocaine 2% (Preservative Free) IO to conscious patients. Syringe bolus (flush) the EZ-IO® catheter with the appropriate amount of normal saline.
- 20-40mg for adult patients
- > 0.5mg / kg for pediatric patients
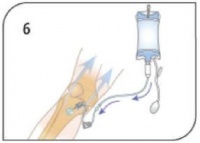
- Utilize pressure (syringe bolus, pressure bag or infusion pump) for continuous infusions where applicable. Begin infusion. Dress site, secure tubing and apply wristband as directed. Monitor EZ-IO® site and patient condition – Remove catheter within 24 hours.
